Arrange the following substances in order of increasing magnitude of lattice energy: KCl, SrO, RbBr, CaO. Ask your peers! Promoted by 23andMe.. Given the lattice energy of kcl = 699 kj,mol, the ionization energy of k = 419 kj,mol and the electron affinity of cl = ?349 kj,mol, calculate the ?
30.12.2014 — For KCL, the melting point is about 1418oF or 770oC, and its lattice energy is about 715 kJ/mol. Recall that energy needed to break apart an .... Solution ) · According to Born – Haber cycle- · Δ H f 0 = ΔH sub + D/2 + IE + EA +U · U = – 719 KJ/mole · Lattice energy of KCl = 719 KJ/mole Ans.
lattice energy
lattice energy, lattice energy trend, lattice energy formula, lattice energy of nacl, lattice energy of mgo, lattice energy of mgcl2, lattice energy of cacl2, lattice energy of kcl, lattice energy of al2o3, lattice energy of na2o, lattice energy is an estimate of the bond, lattice energy definition, lattice energy calculator, lattice energy definition chemistry, lattice energy equation
13.08.2019 — In this article, we will discuss the right half-plane zero, a byproduct of pole splitting, and its effects on stability.. The lattice enthalpy is equal to −x. ... EXAMPLE 4.14 Using a Born–Haber cycle to determine a lattice enthalpy Calculate the lattice enthalpy of KCl(s) .... von C Kittel · Zitiert von: 39493 — The total lattice energy of the crystal of 2N ions at their equilibrium ... Figure 10 Energy per molecule of KCl crystal, showing Madelung and.. vor 2 Tagen — Incoming light can hit the electrons in the semiconductor lattice and move them to a higher energy state, at which point they are free to .... Lattice Energies (E. ... KCl, 698. KBr, 672. KI, 632. AuCl, 1042. AuI, 1050. TlCl, 748. BaF2, 2368. TiF2, 2749. ZnF2, 2971. ZnS, 3619. CdS, 3402. HgS, 3573.

lattice energy formula
lattice energy definition chemistry
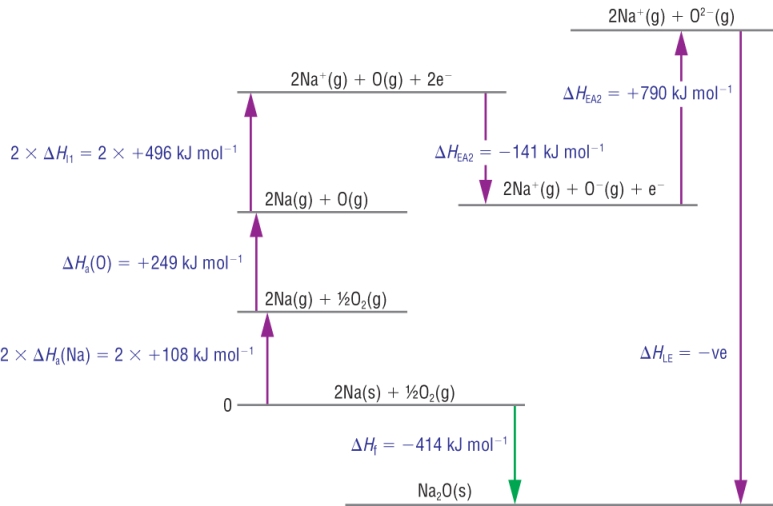
College Chemistry · nah.;. Anonymous. Nov 18, 2010 · Nacl have greatr lattic energy as campard to kcl bcz cl have same but size of Na is small and size of k .... Using Born-Haber cycle for KCl,Substituting the values we have, The reverse of the above equation i.e.Hence the lattice enthalpy of KCl = +719 kJ mol-1.. Why / how is it possible that KCl has a higher melting point but a lower lattice energy when compared to LiCl? This seems counter intuitive. Is …. 01.02.2008 -- Which compound in each of the following pairs of ionic substances has the most exothermic lattice energy? Justify your answers NaCl or KCl.. H Na + (g) + Cl - (g) H lattice energy of formation Na (s) + ½ Cl 2 (g) ... e - + Cl (g) H ionisation energy/ies H electron affinity/ies KCl 90 121 .... von S Froyen · 1986 · Zitiert von: 116 -- Calculated values for the lattice constants and bulk moduli are in good agreement ... Finally the authors have calculated the electronic energy levels at .... 2 CrCl3 + 3 K2CO3 = Cr2 (CO3)3 + 6 KCl. ... Consequently, the Cr3+ ions form a triangle lattice on the twin boundary, as schematically shown in Fig. 4.. 13.05.2019 -- Titration Method:Preparing pure dry crystals of sodium chloride (NaCl) from hydrochloric acid (HCl) and sodium hydroxide (NaOH)Before the .... When it is dissolved in water, its lattice breaks up, the ions become hydrated, and they also disperse ... Similar effects were not found with CrCl3 or KCl.. Hint: Lattice energy: The amount of energy released when \( 1 \)mole of an ionic solid is formed from its gaseous ions.Complete step by step answer:Lattice .... 16.01.2021 -- Lattice Energy & Ionic Bonds: Problem 6.58: Order the following compounds according to their expected lattice energies: LiCl, KCl, KBr, .... Problem 6.58: Order the following compounds according to their expected lattice energies: LiCl, KCl, KBr, MgCl2. The potential energy between two ions is .... Key Points. Lattice energy is defined as the energy required to separate a mole of an ionic solid into gaseous ions. Lattice energy cannot be measured .... 01.11.2020 -- Hi, The enthalpy of crystallization of $KCl$ (Potassium Chloride) is + 715 kJ/mol. The enthalpies of hydration for Potassium and Chloride .... 15.10.2014 -- The lattice energy for KCl is 715 kJ mol-1. ... Which have the lowest lattice energy NaCl KCl KI LiCl? KI .... Lattice energy of kcl ... As defined in Equation (ref'eq1), the lattice energy is positive, because energy is always needed for the ions to blur.. 05.06.2014 -- (i) Br- is bigger than Cl- so KCl has a higher lattice energy and a higher melting point than KBr. (ii) K+ is larger than Na+ so NaCl has a .... von SC O’Hern · Zitiert von: 597 -- reactive defects were first introduced into the graphene lattice through ion ... Upon etching, both the membrane potential and the rate of KCl diffusion .... Kcl lattice energy. Discussion Questions How is lattice energy estimated using Born-Haber cycle? How is lattice energy related to crystal structure?. 28.08.2018 -- Lattice energy is defined as the energy required to separate a mole of an ionic solid into gaseous ions. ... LiCl > NaCl > KCl > RbCl > CsCl.. 29.10.2015 -- Lattice energy formation is the energy released when one mole of a ... K(s) + 1/2Cl2(g) KCl(s) K(g) Cl(g) K+ (g) + Cl- (g) ΔHf= -436 kJ .... Q.) Lattice energy of KCl is measured in kilojoule per mole which is. 1), −690. 2), −900. 3), −890. 4), −560. 5), NULL .... Table shows lattice crystal energy in kJ/mol for selected ion compounds. ... KCl, 701, 720. NaHSe, 703, 732. NaBH4, 703, 694. TlOH, 705, 874.. 5. (a) Calculate a value for the lattice energy of KCl using the Kapustinskii Equation. (b) Calculate the lattice energy for KCl using the data given below.. Born Haber Cycle, Basic Introduction, Lattice Energy, Hess Law \u0026 Enthalpy of Formation - ChemistryHeating a solution of KCl in lab How to use .... The lattice energy of potassium chloride, KCl, is -715 kJ/mol. In which compound is the bonding between ions stronger? Why? - Slader.. Beryllium is a chemical element with the symbol Be and atomic number 4. It is a steel-gray, ... Thus, for high-energy neutrons, beryllium is a neutron multiplier, .... Solution for Which of the following ionic compounds has the greatest lattice energy? 1. NaF 2. KBr 3. CrN 4. KCl 5. NaCl.. The energy released by the attraction between the ions is called the. Lattice Energy. The reaction is... Na. +. (g) + Cl. -.. 900] 9 people answered this MCQ question is the answer among −900 for the mcq Lattice energy of KCl is measured in kilojoule per mole which is.. Rank the following in order of increasing lattice energy. MgO NaCl KCl. Find the charges of the compounds. Mg²⁺ O²⁻ Na¹⁺ Cl¹⁻ K¹⁺ Cl¹⁻. 12.02.2021 -- ... scanning electronic microscopy, and energy dispersion spectrometer. ... The bonding of copper ions to lattice oxygens dictates the .... 26.05.2016 -- The reticular enthalpy is the opposite of this enthalpy variation. 290-CH 3 CH 2 4 NHCl 2. Solved The Lattice Energy Of Potassium Chloride Is .... Lattice formation energy: -7.19 eV. Lattice parameter: 3.14 Å (3.116 at 0K.) Density .... 30.10.2019 -- The heat of solution of KCl is 17.2 kJ/mol and the lattice energy of KCl(s) is 701.2 kJ/mol. Calculate the ... kJ (D) -718 kJ (E) None of .... From the lattice energy of KCl in Table 9.1 and the ionization energy of K and electron affinity of Cl in Tables 8.2 and 8.3, calculate the for the reaction.. Lattice Energies and the Strength of the Ionic Bond. The force of attraction between oppositely charged particles is directly proportional to the product of .... von M Nakayama · 2012 · Zitiert von: 24 -- KCl) were used as the counter and reference electrodes, ... defects in the MnO2 lattice,30 although details to support this have.. 29.02.2016 -- The problem provides you with the lattice enthalpy of potassium chloride, KCl. ΔHlatt=−701 kJ mol−1. When lattice enthalpy carries a .... From the lattice energy of \mathrm{KCl} in Table 9.1 and the ionization energy of \mathrm{K} and electron affinity of \mathrm{Cl} in Tables 8.2 and 8.3, .... Q.1 Which substance in the the following pairs has the larger lattice enthalpy? a) NaCl or KCl b) NaF or NaCl c) MgCl2 or NaCl d) MgO or MgCl2 .... Calculate the lattice energy for lithium fluoride given the following information: ... ionic solids in order of decreasing lattice energy: KCl, LiCl, NaCl.. 01.06.2021 -- matter, material substance that constitutes the observable universe and, together with energy, forms the basis of all objective phenomena.. AStotal > 0 for a spontaneous change ( 17.72 ) 17.11 Enthalpy changes and ... lattice energy of KCl is – 718 kJ mol- ' and so if the dissolution of KCl .... 11.12.2019 -- Get the detailed answer: The lattice energy of KCl is -715 kJ/mol, and the enthalpy of hydration of one mole of gaseous K+ and one mole of .... 3b) (7) Radiative intensity is defined as radiative energy flow per unit ... we could calculate the maximum concentration of KCl that can be absorbed by the .... 24.12.2016 -- Lattice energy depends on the magnitude of the charges of the ions ... Comparing NaCl and KCl, we see that the lattice energies are: 788, .... 07.03.2021 -- Category: Lattice energy of kcl ... Lattice enthalpy and lattice energy are commonly used as if they mean exactly the same thing - you will .... 19.03.2018 -- [image] Rank from largest to smallest magnitude of lattice energy. B) Draw each molecule with their ... of lattice energy.CaO, NaF, SrO, KCl.. 02.06.2021 -- 1 Answer to The heat solution of KCl is 17.2 kj/mol and the lattice energy of KCl (s) is 701.2 KJ/Mol. Calculate the total heat of hydration .... the lattice energy, or lattice enthalpy ΔHG must be provided. ... (LiCl), potassium chloride (KCl) and sodium chloride (NaCl) will.. Solvotrode LiCl, NaCl, KCl, RbCl, and CsCl? ... intermolecular forces between the water molecules is stronger than the bond energy within the LiCl lattice.. von SS De Souza · 1999 -- Computer modelling methods, based on the lattice energy and the defect energy minimisation, have been used to study the possibility of dipolar defect .... Use the born-haber cycle to calculate the lattice energy of kcl(s) given the following data. Discussion Issues How Is the Grid of Energy Evaluated using the .... Chem 2 AP: Homework #9-1: Ionic Bonding, Lattice Energy and Born-Haber Cycle Diagrams ... (a) KCl or MgO: MgO because its charges are twice as big as KCl.. 28.06.2020 -- The lattice energies for the alkali metal halides is therefore ... In respect to this, which has a higher lattice energy KCl or MgO?. von CR Gopikrishnan · 2012 · Zitiert von: 23 -- First principles calculations based on DFT have been performed on crystals of halides (X = F, Cl, Br and I) of alkali metals (M = Li, Na, K, Rb and Cs).. ... of lattice energy and explain how you came up with the answer... KCl, SrO, RbBr,CaO and CsI, KBr, KI, MgS, BeO. SO I get KCl, RbBr, MgO .... Therefore, the lattice energy of CaCl2 is greater than that of KCl. Distance between the Ions. The lattice energy of an ionic compound is inversely proportional .... Question: What Is The Lattice Energy Of KCl? Use The Given Information Below. Heat Of Formation For KCl = -436.7 KJ/mol Heat Of Sublimation For K = 89.20 .... von G Graeffe · 1966 · Zitiert von: 4 -- The activation energies of the steps are nearly equal. NaCl and KCl form a complete range of stable mixed crystals only at high temperatures. The.. Arrange the following in the increasing order of lattice energy LiCl, Nacl, KCl, RbCl, CsCl?Factors affecting lattice energy If inter ionic distance .... The lattice energy of NaCl NaF KCl and RbClfollow the order A KCl RbCl NaCl NaF B NaF NaCl KCl RbCl C RbCl KCl NaCl NaF D NaCl RbCl NaF KCl.. von J Vail · 1973 · Zitiert von: 5 -- ... reorientation of FA(Na) and FA(Li)-centres in KCl are analysed theoretically. ... c) the lattice energy of the defect, and d) the net effect of harmonic .... Mar 25, 2015 · CrCl2 + K2S ----> KCl + CrS all sulfides are insoluble except Alkali metals and Alkaline earth ... K2S (s) + HOH Calculated lattice energies.. Lattice energy of KCl is measured in kilojoule per mole which is −690 −900 −890 −560. Chemistry Objective type Questions and Answers.. 22.05.2017 — KCl의 격자에너지. lattice energy of KCl 다음 자료를 사용하여 염화포타슘의 ΔH°_f를 구하라. K(s) + 1/2 Cl2(g) → KCl(s) 승화 엔탈피, .... these data only, compute the cohesive energy of KCl. ... To get V.0, the lattice energy which includes the core repulsion term. Once we.. 16.01.2021 — Use the Born-Haber cycle to calculate the lattice energy of KCl(s) given the following data: ΔH_{sublimation} (K) = 79.2 kJ/mol IE (K) .... Click here to get an answer to your question ✍️ The lattice energies of NaCl, NaF, KCl and RbCl follow the order:. Lattice enthalpy is an exothermic process as large amounts of energy is released upon the formation of ... K(s) + 1/2Cl2(g) à KCl(s) DH qf = - 437 KJ Mol-1.. von E CATIKER · Zitiert von: 5 — Activation energies of decomposition decreased from 224.14 to 86.125 ... inhibitor and well-dried KCl ([KCl]=[M]/3) were added into the .... Jul 27,2021 - Calculate the lattice energy of KCl at 298 K. Given that:Sublimation energy of K(s) = 89 kJ mol–1Ionization energy of K(g) = 418 kJ .... This page introduces lattice enthalpies (lattice energies) and Born-Haber cycles. Lattice enthalpy and lattice energy are commonly used as if they mean exactly .... 9.3 Lattice energyThe calculation and determination of lattice energyNa + (g) + Cl - (g) NaCl (s) + U(Lattice Energy)Overall electrostatic interaction .... vor 6 Stunden — There might be issues withSNV table fraternalilab.kcl.ac. ... CRYSTAL LATTICE SPACING: 1.92017 STEP NUMBER OFFSET: 0.00 ABbmrb.ioblastp ...
900d8beed2
TГ©lГ©chargement Yoga for beginners apk
Chennai Express Movie Hindi Dubbed Mp4 Hd Download
Developer Assistant_v1.1.0(2001132112).apk
Denon Dct A100 Manual Woodworkers
Howie Day, Stop All The World Now (Special Edition) Full Album Zip
chapter 8.1 atp adp energy cycle worksheet answers
Hindi dubbing audio track Deadpool (2016) AC3 В« Audio Tracks for Movies
pink friday deluxe edition download zip
6th order bandpass
Heroclix Maps Download Free
